What you see when you spray out a finish, are the atomized particles of finish forming a wet spot on the surface of the wood. As the solvents evaporate, the coating lays out into a flat film. This observation is only a small part of what is actually going on, as the pictures above demonstrate. What happens between the time you pull the trigger and the coating dries is truly what finishing is all about.
Fundamentals
The fundamentals of wood finishing involve principles that occur at a microscopic level. We can not see them with the naked eye so we tend to just accept the results without understanding why they happen. This leads to many of the common problems that we have and the frustrations that follow. Many of us react to the problem without really understanding the process that causes it. Because the physical properties of water are different than the solvents we are used to, it is necessary to expand the description of the process.
Properties of Water
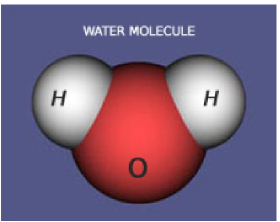
Water is a combination of two hydrogen atoms and one oxygen atom. The atoms are arranged in a triangular fashion. The oxygen atom has a slightly negative charge and the hydrogen atoms have a slight positive charge. As with magnets positives and negatives, called dipoles, attract each other.
These are some of the physical properties of water as they relate to waterborne finishes:
- Cohesion is the attractive force between molecules. Water binds to itself very easily. Due to the triangular arrangement of the hydrogen atoms, and the dipolar attraction of the positive and negative charge on the hydrogen and oxygen atoms, when one water molecule comes in contact with another water molecule they stick to each other. This is why water will bead up on the waxed surface of your car. This is also why waterbornes will not adhere to oily fingerprints on a piece of wood.
- Surface Tension is the force created by cohesion. Surface tension is defined as the attractive property of the surface of a liquid. It causes many liquids to behave as elastic sheets. This is what allows objects to float on liquid surfaces and causes liquids to form droplets. This is also what makes the water drops round over causing them to cover only the smallest possible surface area. Water has a higher surface tension than most organic solvents. Only mercury has a higher surface tension than water. Surface tension is the force that must be overcome when atomizing waterborne finishes.
- Adhesion is the attractive force between molecules of different types. Water sticks very well to other surfaces. When you pour water onto the waxed surface of a car, it will bead up and stick to the paint even on non-horizontal surfaces. The adhesive forces are greater than the cohesive forces so the water sticks to the paint rather than to itself.
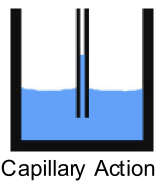
- Capillary Action is the ability to move up a narrow-diameter tube. Capillary action is also what allows water to move up the hollow fibers of plants and trees. The water adheres to the inside of the fiber, but surface tension tries to flatten it out. This makes the water rise and bind to itself again. This process continues until it can no longer overcome the force of gravity. Capillary action also draws the water into the fibers of the wood when you spray, and causes grain raise.
- Specific Heat is the amount of heat energy that is required to raise the temperature of a liquid. Water has a very high specific heat so it takes a lot of energy to break the hydrogen bonding between molecules which allows it to evaporate. This is why waterborne dries slower than solvent-based.
- Solvent – water is a strong solvent, considered to be a universal solvent. It will dissolve many types of substances and turn them into a solution. Unfortunately, water does not dissolve the polymer resins in waterborne finishes.
- Miscible is the ability to mix with another liquid, in any proportion and form a single homogenous liquid. Water is mixed with ethyl glycol, another solvent to form the solvent base of waterborne finishes, which is a good thing. Water is completely miscible with air and that often can lead to problems in the finishing world.
- Positive Azeotrope – water mixed with certain liquids creates a solution that has a lower boiling point than either of the two original liquids. For waterborne finishes, this is the coalescing solvent. The reduction in the boiling point reduces the minimum film-forming temperature of the polymer in the finish and helps decrease dry time
- Water is transparent, and in small quantities appears to be clear. In reality, water is a very light blue