
When you pull the trigger on your spray gun, a stream of material exits the nozzle. The pressure behind the fluid, as well as the tiny holes next to the large center hole in the air cap, flatten the stream into a sheet. As the liquid continues to move forward the sheet disintegrates into spaghetti-like ligaments. The forward momentum finally overcomes the surface tension of the liquid and droplets form. As the droplets move forward they fan out in a cone shape. The air coming out of the air cap horns controls the final shape and size of the fan.
This process is a balancing act between:
- The amount of fluid that you set to come out of the gun
- The amount of air pressure that you supply
- The size of the nozzle
- The size and placement of the holes in the air cap
- The viscosity of the finish
- The surface tension of the liquid you are spraying.
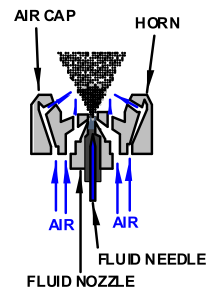
We can control the viscosity of the finish and the settings on the gun. We do this to accommodate the nozzle/air cap combination to give us the desired atomization.
When thinking about waterborne finishes, you have to remember that the major component in the finish is water, usually 60% or more. Solvent-based finishes contain typically 60-to-80% organic solvent. Since water has a higher surface tension than most organic solvents, we need to adjust our settings and/or reconfigure our equipment to overcome the surface tension of the water. This difference in molecular attraction is what causes a lot of problems when people switch from solvent-based to waterborne products. Reaction to temperature, viscosity, reduction, and application techniques are all just a little bit different than what most finishers are used to. The solutions are not difficult; they just require a different mindset. A “waterborne mentality”, so to speak.
After the finish is applied to the wood, the water and coalescing solvents are removed from the pool of liquid by evaporation. Since the coalescing solvent in the finish is selected, it evaporates slower than water. As it evaporates, the water below the surface migrates to the surface by diffusion. Diffusion is defined as “the spontaneous migration of substances from regions where their concentration is high to regions where their concentrations are low”. For example, when you bake cookies in the kitchen and smell travels all over the house. This is diffusion.
This flow of water to the surface continues until it is evaporated. The time that it takes for the water to evaporate is based on:
- The thickness of the coating
- The percentage of water in it
The evaporation rate is determined by:
- The type and amount of coalescing solvent
- The temperature of the air
- The humidity of the air
- The velocity of the air moving over the surface
- The temperature of the coating.
Temperature
The acceptable temperature and humidity range for the application of waterborne finishes is a little narrower than solvent-based finishes.
The air, wood, and finish should all be above 65 degrees when spraying. Low temperatures increase dry time and increase the potential for trapping dust. They also raise the viscosity of the finish which raises surface tension.
This increase in surface tension accentuates the pores of open-grained woods because the finish clings to itself and won’t flow onto the pore.
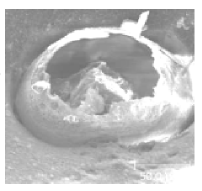
Temperatures that are too low or too high can cause an increase in pinholes. The air that atomizes the liquid finish and solvents needs to go somewhere when the finish is sprayed on the wood. Microscopically, they form tiny bubbles and float to the surface. In ideal conditions, the bubbles pop at the surface and the liquid simply flows back and fills in the little pinholes. When the temperature is low and the viscosity is high the bubbles take a lot longer to float to the surface. When they finally get to the surface and pop, the liquid is so thick that it often doesn’t flow back into the hole causing the pinhole. This problem is often exacerbated by the air pressure increase from atomization, thus creating more bubbles. The solution to this problem is simple: Either increase the temperature of the water to reduce the viscosity, or apply less wet mils.

When temperatures are too low, the coalescent solvent evaporates before the resin levels and hardens. This results in an orange peel. There are flow agents that can be added to the finish that can help with this problem.
High temperatures also can cause pinhole problems. The bubbles move quickly to the surface after being sprayed, but the higher temperatures also cause the surface of the finish to dry quickly. This raises the viscosity at the surface which impedes the flow of the liquid back into the hole; again pinholes are the result. The solution is to add some flow agent which will keep the surface wet longer and allow the pinholes to heal.

Another problem in high-temperature conditions is the finish drying too fast, trapping the water. The result is that the finish looks hazy especially over dark stains. This is because the water in the coating must evaporate before the film can form.
Some waterborne finishes are not tolerant of freezing, which can cause resins to coagulate. This causes the finish to no longer be an emulsion. For this reason, waterbornes must be stored in heated buildings. This can also become problematic when shipping certain products to the northern part of the country or Canada, in the wintertime. Some manufacturers ship their products in temperature-controlled trucks.
Humidity
For the finish to dry and form a film, the water in the finish must evaporate into the air. As stated earlier, temperature and relative humidity are two of the major factors that influence the evaporation rate. Relative humidity is a measure of how much water vapor is in the air at a given temperature.

The relative humidity fluctuates with air temperature. As the temperature increases so does the water-holding capacity of the air. For example, if an area is 77°F with 50% relative humidity, a 10% rise in temperature will increase the potential water capacity of the air by about 40%. Increased temperature increases evaporation and the increased potential for the air to absorb water vapor results in shorter dry times. That is the good part. The bad part is that at higher temperatures, the ambient air may already be heavily loaded with water vapor due to the environmental conditions at the time.
The quality of your compressed air is especially important with waterbornes. When the relative humidity is high it is very important to monitor your air filters and compressor for excessive water. Draining your compressor and filters regularly will keep the water from being passed on and contaminating your finish.
Grain Raise
Waterborne raises the grain when applied; this goes back to the capillary properties of water. This is often addressed by sanding the surfaces to finer grits, almost burnishing the wood. Wood that is sanded with 120 grit has more surface area of wood fiber exposed to the liquid than wood sanded to 240 grit. I have tried lightly wetting the wood surface before sanding and then knocking down the raised grain, but I’ve had mixed results that did not seem to warrant the extra step.

If you apply a heavy seal coat, it will stay wet on the surface longer and increase the chances of grain raise. Heavy coats can also trap water below the surface of the film as it dries resulting in a hazy appearance. This is particularly noticeable over dark stains. Apply several light coats before sanding the sealer coat. To avoid intercoat adhesion problems, a wet-on-wet application requires that the successive coats be applied within minutes of each other, especially in warm weather when dry time is shortened. This procedure will not only reduce overall dry time, but will reduce grain raise as well. The application of two light coats before sanding will reduce the risk of burn-throughs.
Make sure that the wood is warm when the finish is applied. If the wood is cool – again, it will slow down evaporation and allow the surface to be wet longer which increases grain raise. If the finish is warm and the wood is cool, there is the chance for water entrapment in the dry film which will result in a hazy finish.

I have found it very difficult to use waterborne primers on MDF. It seems no matter how fine I sanded the bare MDF, or how lightly I applied the primer, the fibers of the MDF keep popping up. When you sand the primer, you re-expose the raised fiber and it again expands when re-wetted with the next coat. The best solution would be to apply a shellac-based primer followed by a waterborne paint.
Softwoods such as pine can also be problematic. Their grain structure is very different than that of

most hardwoods. Most pines have a larger open cell structure so they are much more porous. Thus, they will absorb more water which results in increased grain raise.
PH
PH levels of the wood are important. If the wood is too acidic, it will impede film formation and lower hardness and chemical resistance. Occasionally this can be a problem with woods that have high

tannic acid content like oak. On painted wood, the tannins can bleed through the paint and discolor it. Wood that has been stripped with an alkaline-based stripper can also cause these problems.

Rust
The water in waterborne finishes will cause ferrous metals such as steel to rust. It can also cause aluminum to oxidize. Most professional spray guns have their wetted parts fabricated from stainless steel or plastic to eliminate this problem. If you are using a pressure pot that is not stainless steel, use a plastic liner and be sure that your pick-up tube is stainless steel. Waterborne products should also be stored in stainless steel or plastic containers.
Never use steel wool on the wood or finish. Any leftover particles will react with the water and will cause black stains in the wood or finish.
Finishing Environment
Contamination
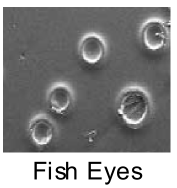
In any finish room, cleanliness is the rule. A clean environment and clean hands are a must. Clean, compressed air is imperative. As the saying goes: Oil and water don’t mix. Check or replace your filters and elements. If your compressor is passing oil determine the cause and fix it. Ignoring these precautions will result in fish eyes or craters.
Avoid the use of tack cloths. Some can leave an oily residue that the finish will not stick to or can cause fish eyes. It is better to wipe the surface with a dry or slightly damp rag.
Health and Safety
Though not very flammable, air quality is still an issue. Spray finishing produces mists and these mists contain resins that can clog or irritate the lungs. Waterborne finishes also contain 2-10% organic solvents which are coalescing solvents.

Depending on the solvent, they may or may not be on the EPA’s Hazardous Air Pollutants (HAPS) list. While the solvents may be encapsulated in water when they evaporate (theoretically making them less harmful), a solvent is a solvent. What may not be on a list today may be on one tomorrow. Wear a respirator and have adequate ventilation. Remember too that the overspray is flammable, so be sure to practice good housekeeping practices. Do not be lulled into complacency when it comes to your health. Contact your finish manufacturer for specifics about their products.
